Dear friends and colleagues,
As a careful optimist, I base my dreams on positive thoughts but my decisions on data.
Vaccines are coming and initial results look promising. Pfizer announced that its vaccine efficacy jumped to 95%, which is now very comparable with Moderna’s.
*Disclaimer*: Clinical trials are evolving, this article reflects interim results as of Nov 18, 2020. Note that safety and efficacy data have not been fully released.
#1. When will I be able to get vaccination and end this nightmare?
This is the question I get the most, so I defer to Dr. Fauci’s latest view
“I would say starting in April, May, June, July — as we get into the late spring and early summer — that people in the so-called general population…could get shots”, said Dr. Fauci.
If both vaccines are authorized (hopefully end of month)…
Government officials anticipate having enough doses for 20 million people by end of 2020
General guidelines from governments, although specific states may vary
Phase 1:
Healthcare workers & first responders
Older adults with underlying conditions
Phase 2:
Teachers & essential workers
People with underlying conditions
Phase 3:
Healthy adults
Children
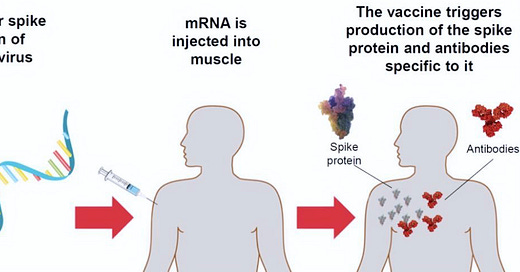
#2. How does mRNA vaccines work in the body:
Both Pfizer and Moderna vaccines use similar mechanism of action: mRNA for coronavirus spike proteins. The mRNA is the genetic material which encodes for the “spike protein” receptor portion of the coronavirus. It is injected into the body and the spike proteins reproduce and attach to human cells ( kinda like isolated claws of an eagle). Once the spike protein is attached, your body produces an immune response to protect you.
*Key things to know*:
Both Pfizer and Moderna mRNA-based vaccines do *not* contain the virus itself. Therefore, the vaccine cannot give you COVID-19.
Both prevent COVID-19 symptoms, but it is uncertain whether it prevents actual infection.
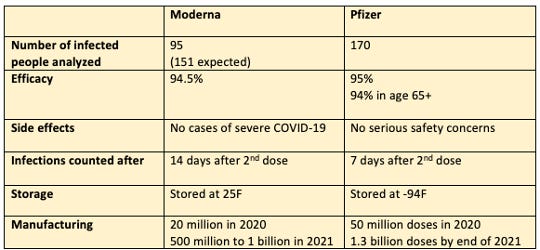
#3. How to think about Moderna vs. Pfizer vaccines:
First of all, how do you interpret the 95% effectiveness?
Take Moderna for example, they studied 30,000+ people. 95 people ended up naturally getting COVID. Out of these 95 infected, 90 people were in the placebo group, and 5 were in the vaccine group. 90 placebo divide by 95 total = 94.7% prevented by vaccine.
Note both vaccines the 95% efficacy data shown are based on 2 doses. Meaning a person would take a first dose, wait an amount of time, take another dose.
What about safety?
Safety effects were tested in the 30,000-40,000 population and no serious adverse effects were observed. Minor side effects include injection site pain, fatigue, muscle pain, etc. However, long-term side effects are not yet determined.
Key difference between the vaccines:
Because mRNA is basically genetic material, it can be very unstable. Currently Pfizer’s vaccine will need to be stored at -94F which means it would require very cold containers for shipping. Moderna claims its vaccine can be maintained at 25F, which medical freezer temperatures can maintain.
Vaccines don’t save lives. The act of vaccination does. This requires robust manufacturing, logistics, transportation, and vaccination programs & education. We will look into how the public and private sector will work together in the next few months…but most importantly, stay informed on facts.
Stay brave,
Kath
P.S. On Thursdays, I will try to publish a concise summary on a healthcare topic for 30 Seconds Healthcare. If anyone has ideas for next Thursday, let me know.
Direct sources
Pfizer: https://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-conclude-phase-3-study-covid-19-vaccine